“Our research lies at the interface of development, ecology, and evolution. We use experimental and comparative methods, guided by mathematical modelling and conceptual analysis”
- Plasticity and Evolution
- Extra-Genetic Inheritance
- Causes and Consequences of Hybridization
- Adaptation in Alien Species
- Ecology and Evolution of Social Complexity
- Extending the Evolutionary Synthesis
Plasticity and Evolution
Organisms are inherently responsive to their environment. They need to be to develop and maintain biological functions. We study how environmental responsiveness evolves and – when it has evolved – how this affects the capacity for organisms to undergo future evolution.
Phenotypic plasticity is everywhere. But when will it be the best solution, what should the response be like, and how long should individuals stay open to environmental influence? Plasticity may also affect the capacity of populations to evolve since it influences the phenotypic variation exposed to selection. To understand the evolutionary implications of plasticity it is often useful to think of phenotypes in terms of regulatory developmental systems. This allows us to study how environmental control evolves, and how this influences how accessible new phenotypic variants are through mutation.
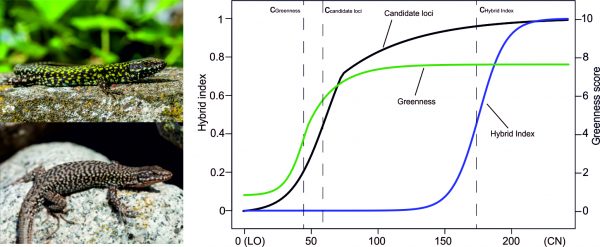
 One prediction is that plastic responses – as a form of developmental bias – can direct adaptive evolution along particular trajectories. This, in turn, may influence what adaptive solutions will look like, and therefore evolutionary convergence and divergence. We test these predictions using a range of methods, from comparative analyses to experimental studies of lizards and other organisms.
One prediction is that plastic responses – as a form of developmental bias – can direct adaptive evolution along particular trajectories. This, in turn, may influence what adaptive solutions will look like, and therefore evolutionary convergence and divergence. We test these predictions using a range of methods, from comparative analyses to experimental studies of lizards and other organisms.
If you want to know more:
Uller, T., Moczek, A.P., Watson, R.A., Brakefield, P.M. & Laland, K.N. 2018. Developmental Bias and Evolution: A Regulatory Network Perspective. Genetics 209: 949-966.
English, S., Fawcett, T.W., Higginson, A.D., Trimmer, P. & Uller, T. 2016. Adaptive use of information during growth can explain long-term effects of early-life experiences. American Naturalist 187: 620-632.
Laland, K.L., Uller, T, Feldman, M., Sterelny, K., Müller, G.B., Moczek, A., Jablonka, E. & Odling-Smee, J. 2015. The extended evolutionary synthesis: its structure, core assumptions, and predictions. Proceedings of the Royal Society of London 282: 20151019.
Pen, I., Uller, T., Feldmeyer, B., Harts, A., While, G. M. & Wapstra, E. 2010. Climate-driven population divergence in sex-determining systems. Nature 468: 436-439.
Badyaev, A. V & Uller, T. 2009. Parental effects in ecology and evolution: Mechanisms, processes, and implications. Philosophical Transactions of the Royal Society, 364: 1169-1177.
Evolutionary Causes and Consequences of Extra-Genetic Inheritance
Parents pass on a wide range of developmental resources to their offspring through the egg, the placenta and via postnatal provisioning. Behavioral interactions are also important, with social learning being common in both invertebrates and vertebrates. While the transfer of DNA is known as genetic inheritance, other mechanisms that influence parent-offspring resemblance are collectively known as extra-genetic inheritance.
Much of our research has focused on how extra-genetic inheritance evolves, in particular when it provides a flexible means to pass on information between generations. For example, we have shown that incomplete epigenetic resetting between generations can be favored by natural selection if environments show a modest degree of variability and uncertainty. These and other model predictions can be tested empirically, which we do using field and laboratory experiments on water fleas and in studies of the evolution of parental care in lizards.

Extra-genetic inheritance also serves as a source of phenotypic variation that – similarly to within-generation plasticity – can influence if and how populations adapt. Stress-induced parental effects may, over evolutionary time, be converted into phenotypes that are reliably inherited between generations. We are interested in trying to reveal signatures of this process in populations that have recently adapted to new environments, and our research involves both water fleas and lizards.
If you want to know more:
Uller, T. & Helanterä, H. 2017. Heredity in evolutionary theory. In Challenging the Modern Synthesis: Adaptation, Development & Inheritance (eds. P. Huneman & D. Walsh), Oxford University Press
Uller, T., English, S. & Pen, I. 2015. When does natural selection favour incomplete epigenetic resetting in germ cells? Proceedings of the Royal Society of London B 282: 20150682
English, S., Pen, I.R., Shea, N. & Uller, T. 2015. The information value of non-genetic inheritance in plants and insects. PLoS One 10(1): e0116996
Uller, T., Nakagawa, S. & English, S. 2013. Weak evidence for anticipatory parental effects in plants and animals. Journal of Evolutionary Biology 26:2161-2170
Badyaev, A. V & Uller, T. 2009. Parental effects in ecology and evolution: Mechanisms, processes, and implications. Philosophical Transactions of the Royal Society, 364: 1169-1177
Causes and Consequences of Hybridization – from Behaviour to Evolution
How new characters originate and evolve is one of the outstanding problems in evolutionary biology. Recent genomic data suggests that characters that evolve within one lineage can sometimes spread to other populations with limited genetic exchange, resulting in asymmetric introgression and a mosaic genome in the receiving population. However, only rarely are systems sufficiently well understood to predict the degree and direction of hybridization, which limits our ability to interpret genomic signatures of introgression.
Male common wall lizards in western Italy exhibit exaggeration of a suite of sexually selected characters, including morphology, colouration and behavior. This allow them to outcompete males from western Europe that lack these characters. We have shown that this results in asymmetric hybridization and adaptive introgression of the suite of characters following secondary contact in both the native and the non-native range of the species.
Our current research, funded by the Swedish Research Council, is extending this work in several directions. Our aims are to test if genomic and developmental modularity facilitated introgression of this suite of characters, and to identify what – if anything – that limits introgression and how repeatable this evolutionary process is across different contact zones.
If you want to know more:
Yang, W., While, G.M., Laakkonen, H., Sacchi, R., Zuffi, M.A.L., Scali, S., Salvi, D. & Uller, T. 2018. Genomic evidence for asymmetric introgression by sexual selection in the common wall lizard. Mol Ecol, in press
MacGregor, H.E.A., Lewandowsky, R.A.M., d’Ettorre, P., Leroy, C., Davies, N.W., While, G.M. & Uller, T. 2017. Chemical communication, sexual selection, and introgression in wall lizards. Evolution 71:2327-2343
MacGregor, H.E.A., While, G.M., Barrett, J., Perez I de Lanuza, G., Carazo, P., Michaelides, S. & Uller, T. 2016. Experimental contact zones reveal causes and targets of sexual selection in hybridizing lizards. Functional Ecology 31: 742-752
Heathcote, R., MacGregor, H., Sciberras, J., While, G.M., D’Ettorre, P. & Uller, T. Male behaviour drives assortative reproduction during the initial stage of secondary contact in lizards. Journal of Evolutionary Biology 29: 1003-1015
While, G.M., Michaelides, S., Heathcote, R., MacGregor, H., Zajac, N., Beninde, J., Perez I de Lanuza, G., Carazo, P., Sacchi, R., Zuffi, M.A.L., Horvathova, T., Fresnillo, B., Schulte, U., Veith, M., Hochkirch, A. & Uller, T. Sexual selection drives asymmetric introgression in wall lizards. Ecology Letters 18: 1366-1375
Adaptation in Alien Species
 Introduced populations are useful to study evolution. The common wall lizard is native to southern Europe but has frequently been introduced by humans to northern Europe. We have studied the origin of non-native populations in England and how their introduction history has shaped their genetic and phenotypic diversity.
Introduced populations are useful to study evolution. The common wall lizard is native to southern Europe but has frequently been introduced by humans to northern Europe. We have studied the origin of non-native populations in England and how their introduction history has shaped their genetic and phenotypic diversity.
The small population size of these populations should limit their adaptive potential. However, lizards of both French and Italian origin have taken a small step towards viviparity since their introduction to England. This reduces the time needed to complete embryogenesis in the nest. Non-native embryos also develop faster at low temperature than native embryos. These adaptive responses allow lizards to hatch before autumn despite the cool soil temperature in England, and help populations persist north of their natural range.
The evolution of prolonged egg retention and faster developmental rate in non-native populations of both French and Italian origin provide opportunities to study the developmental basis of convergent evolution. By profiling gene expression in early embryos, we have revealed that populations of French and Italian origin have recruited different genes to speed up development, but that the function of these genes are broadly similar. We are now interested in asking what the underlying mechanisms are, if these are similar to those underlying climatic adaptation during natural range expansion, and if maternal effects can facilitate rapid adaptation.
If you want to know more:
Feiner, N., Rago, A., While, G.M. & Uller, T. 2017. Signatures of selection in embryonic transcriptomes of lizards adapting in parallel to cool climate. Evolution, in press DOI: 10.1111/evo.13397
Michaelides, S., While, G.M., Zajac, N., Aubret, F., Calsbeek, B., Sacchi, R., Zuffi, M.A.L. & Uller, T. 2016. Loss of genetic diversity and increased embryonic mortality in non-native lizards. Molecular Ecology 15: 4113–4125
While, G.M., Williamson, J., Prescott, G., Horvathova, T., Fresnillo, B., Beeton, N.J., Halliwell, B., Michaelides, S. & Uller, T. Adaptive responses to cool climate promotes persistence of a non-native lizard. Proceedings of the Royal Society of London B 282: 20142638
Michaelides, S., While, G.M., Zajac, N. & Uller, T. Widespread primary, but geographically restricted secondary, human introductions of wall lizards, Podarcis muralis. Molecular Ecology 24: 2702-2717
Ecology and Evolution of Social Complexity in Lizards
When we think of social animals our thoughts are typically drawn to ants, meerkats, or humans. In contrast, the social life of lizards may seem dull. Not so. Some lizards live in social groups that differ in size and complexity in fascinating ways. Together with Geoff While at the University of Tasmania we study links between behavior and social structure in a group of lizards known as the Egernia.

Our main study species Liopholis whitii lives in family groups. Geoff has shown that the composition and stability of these groups are dictated by both the genetic structure of the population and the ecological conditions. Together these factors shape selection on a suite of characters that are important for understanding the transition to social life.
While family life remains simple in lizards, there are more evolutionary transitions to family groups in lizards than in birds and mammals. Even within the Egernia, species differ in their social systems, ranging from solitary species to those that live in large groups with several overlapping generations. This makes it possible to reconstruct the steps towards social complexity, unravelling how social systems originate and are maintained. Across lizards and snakes, it turns out that live bearing facilitates transitions to sociality, presumably because it promotes interactions between close relatives. Our research on lizard sociality is funded by the Australian Research Council.
If you want to know more:
Halliwell, B., Uller, T., Holland, B.R. & While, G.M. 2017. Live bearing promotes the evolution of sociality in reptiles. Nature Communications 2030
While, G.M., Chapple, D.G., Gardner, M.G., Uller, T. & Whiting, M.J. 2015. Egernia lizards. Current Biology 25:R593-595
While, G.M., Uller, T., Bordogna, G. & Wapstra, E. Promiscuity resolves constraints imposed by population viscosity. Molecular Ecology 23: 721-732
Extending the Evolutionary Synthesis
The 20th century became known as the century of the gene. The overwhelming focus on genes makes it easy to forget that what is outside of the genome is not only permissive for development, but also instructive. Organisms are inherently flexible and respond to environmental challenges by changing their shape, size, or behaviour. Sometimes such responses can even affect the next generation. The evolutionary implications of developmental plasticity and extra-genetic inheritance are poorly understood and highly contested. The fundamental source of contention is if plasticity and extra-genetic inheritance are best viewed as adaptive features under genetic control, or if there is value in considering the processes of development and heredity to play a more active role in the origin, spread, and maintenance of adaptations.
The latter perspective is not new (in fact, it is as old as evolutionary biology itself), but it is gaining popularity through what has become calls for ‘extending’ the evolutionary synthesis. Yet, it is unclear how these perspectives on evolution differs from the gene-centric focus that has been dominant for the last century. To bring clarity to these issues – and perhaps ultimately bring about conceptual change – biologists with different expertise need to work together with philosophers and historians of biology.
Ultimately, conceptual frameworks should be evaluated on the basis of their ability to generate useful research. Much of the work in our group is therefore motivated by putting our ‘work-in-progress’ version view of an extended evolutionary synthesis to the test. This includes a large international effort coordinated by Tobias and Kevin Laland. The initiative, funded by the John Templeton Foundation, include 22 projects and over 50 scientists, philosophers and historians centered at eight institutions, including Lund, St Andrews, Cambridge and Stanford.
If you want to know more:
Uller, T. & Helanterä, H. Niche construction and conceptual change in evolutionary biology. British Journal for the Philosophy of Science, in press, https://doi.org/10.1093/bjps/axx050
Uller, T. & Helanterä, H. 2015. Heredity in evolutionary theory. In Challenges to Evolutionary Biology: Development and Heredity (eds. P. Huneman & D. Walsh), Oxford University Press, forthcoming
Laland, K.N., Uller, T., Feldman, M.W., Sterelny, K., Müller, G.B., Moczek, A., Jablonka, E. & Odling-Smee, J. 2014. Does evolutionary theory need a rethink? Nature 514: 162-165
Laland, K.L., Uller, T, Feldman, M., Sterelny, K., Müller, G.B., Moczek, A., Jablonka, E. & Odling-Smee, J. 2015. The extended evolutionary synthesis: its structure, core assumptions, and predictions. Proceedings of the Royal Society of London 282: 20151019
Laland, K.N., Sterelny, K., Odling-Smee, J., Hoppitt, W. & Uller, T. 2011. Cause and effect in biology revisited: Is Mayr’s proximate-ultimate distinction still useful? Science 334:1512-1516